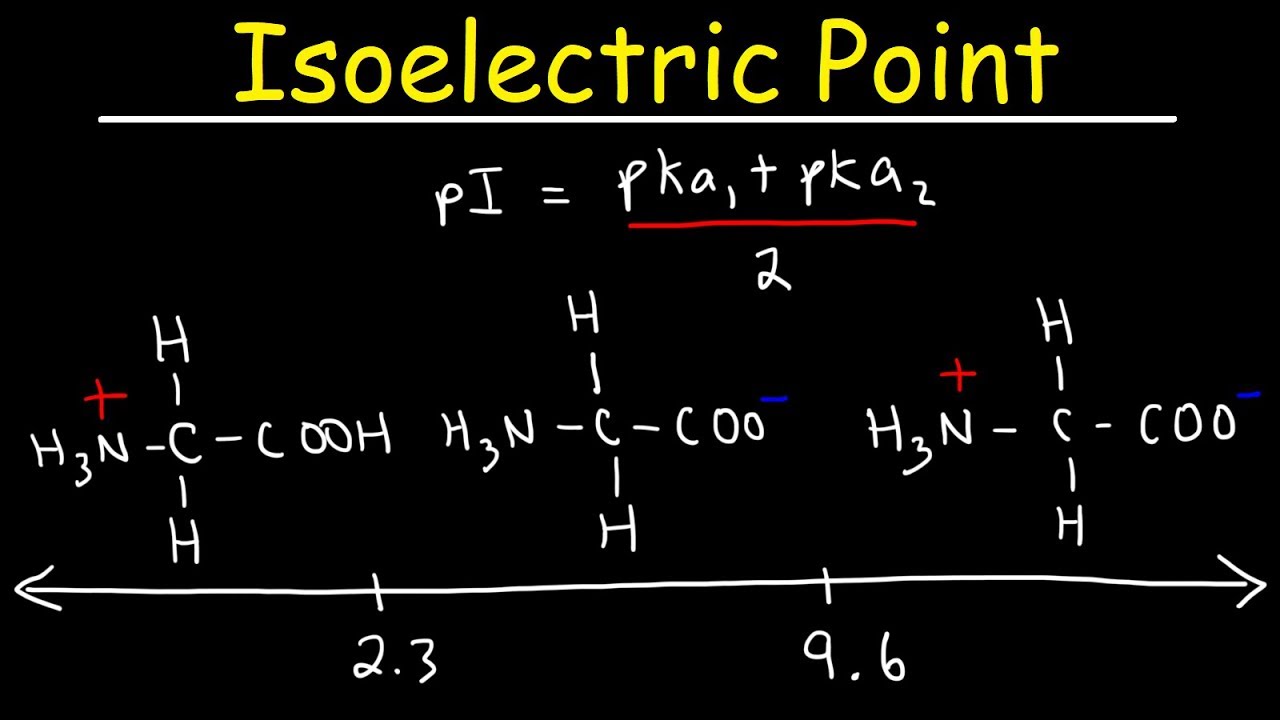
 Thus, the solution has to be acidic to protonate the carboxylate group in the side chain. To balance the negative charge in the side chain, carboxylate ion has to be protonated. This is because at pH 7 the acidic amino acid’s side chain carboxylic acid group exists as a carboxylate ion, so it has a net negative charge. Amino acids having acidic side chains have isoelectric points less than 7.The isoelectric point of neutral amino acids is near pH 7.The isoelectric point of each amino acid is different due to the presence of a unique side chain group, usually represented with the letter R. Sidechain groups present in an amino acid determine the isoelectric point of that amino acid.The isoelectric point of amino acids differs based on the following factors: PI=8.95+10.532=9.74 Isoelectric points of acidic and basic amino acids To calculate the isoelectric point of Lysine, the pKa of the similarly ionizable groups will be considered which are the ammonium group (pKa = 8.95) and the basic side chain (pKa = 10.53). 
Lysine has a basic group in its side chain.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
